January 2016 – The CenterWatch Monthly : PDF
Product Details
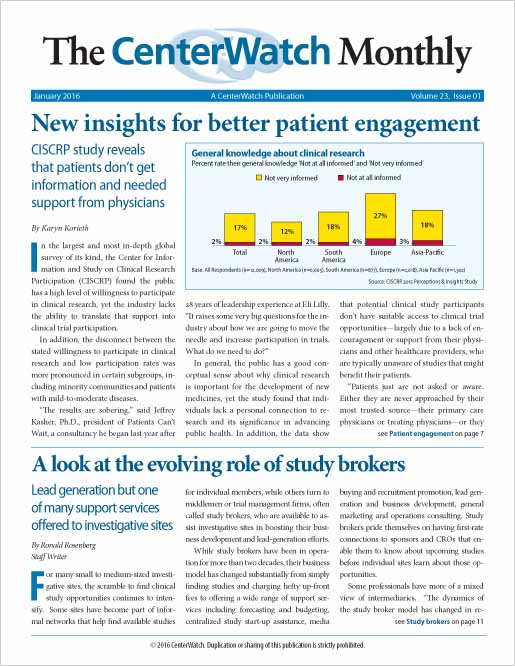
New insights for better patient engagement
In the largest and most in-depth global survey of its kind, the Center for Information and Study on Clinical Research Participation (CISCRP) found the public has a high level of willingness to participate in clinical research, yet the industry lacks the ability to translate that support into clinical trial participation.
A look at the evolving role of study brokers
For many small to medium-sized investigative sites, the scramble to find clinical study opportunities continues to intensify. Some sites have become part of informal networks that help find available studies for individual members, while others turn to middlemen or trial management firms, often called study brokers, who are available to assist investigative sites in boosting their business development and lead-generation efforts.
Also in this issue:
- Understanding feasibility for faster clinical trials
- Engaging advocacy groups for patient recruitment
- Regulatory Update
- Month in Review
- FDA Actions
- Study Lead Opportunities
- New Drugs in the Pipeline