October 2011 – The CenterWatch Monthly : PDF
Product Details
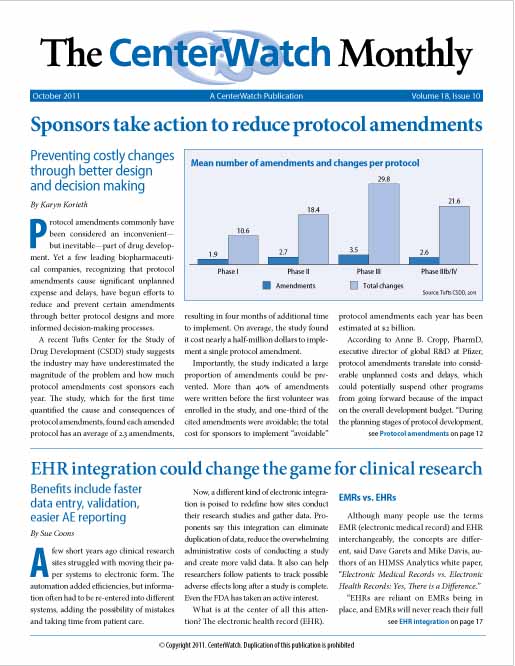
Sponsors take action to reduce protocol amendments
Protocol amendments commonly have been considered an inconvenient—but inevitable—part of drug development. Yet a few leading biopharmaceutical companies, recognizing that protocol amendments cause significant unplanned expense and delays, have begun efforts to reduce and prevent certain amendments through better protocol designs and more informed decision-making processes.
EHR integration could change the game for clinical research
A few short years ago clinical research sites struggled with moving their paper systems to electronic form. The automation added efficiencies, but information often had to be re-entered into different systems, adding the possibility of mistakes and taking time from patient care.
Eye On Isis Pharmaceuticals
Isis Pharmaceuticals focuses on antisense drug development, applicable to many different disease targets.
- Essential steps to Facebook success
- Better management of mobile devices in protocols
- Industry Briefs
- The Pulse on Recruitment
- TrialWatch
- New Study Launches